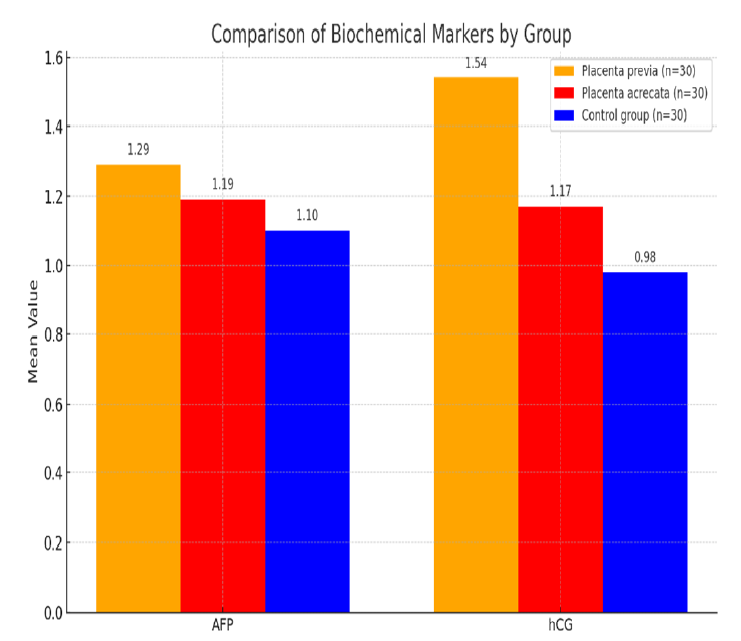
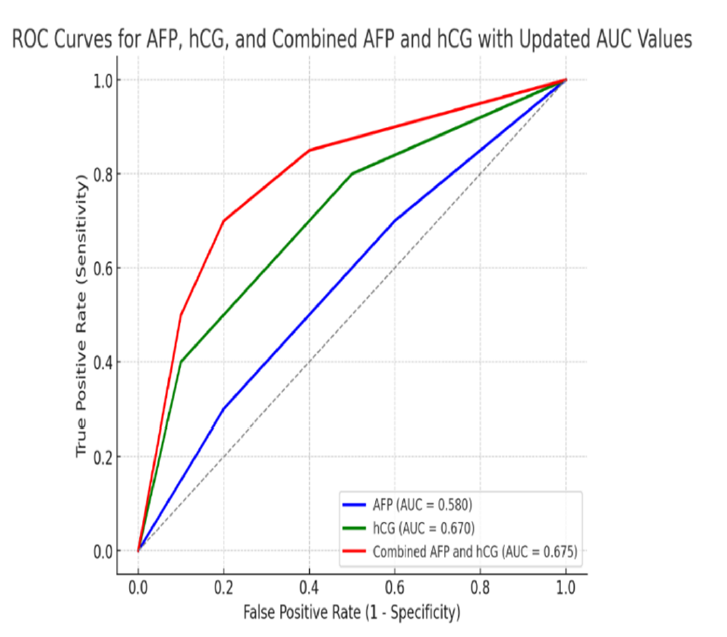
Pathological placentation represents a major obstetric challenge and is a leading cause of maternal and perinatal morbidity and mortality worldwide. Normal placentation depends on tightly regulated trophoblast differentiation, invasion, vascular remodeling, and placental endocrine activity. Disruption of these processes can result in a spectrum of placental disorders, including placenta previa, placenta accreta spectrum (PAS), placental abruption, fetal growth restriction, and hypertensive disorders of pregnancy. Early identification of pregnancies at risk for abnormal placentation remains a critical objective in modern obstetrics, as timely diagnosis allows appropriate surveillance, referral, and peripartum planning to reduce adverse outcomes [1-3]. The human placenta is a discoidal organ that facilitates maternal–fetal exchange through highly specialized villous structures bathed in maternal blood. Its functional unit consists of chorionic villi lined by trophoblast, forming a haemomonochorial barrier that allows efficient transport of gases, nutrients, hormones, and waste products [1-2]. Placental development begins in early gestation with trophoblast differentiation and invasion into the decidua, followed by formation of villous trees and establishment of uteroplacental circulation. During the first trimester, histiotrophic nutrition predominates, while from the second trimester onward, haemotrophic exchange becomes the primary mode of fetal support [3-4]. Any disturbance in these developmental stages may impair placental implantation or vascular remodeling, predisposing to pathological placentation. The placenta is not merely a passive exchange interface but an active endocrine and metabolic organ. It consumes a substantial proportion of maternal oxygen and glucose delivery and produces a wide array of hormones essential for pregnancy maintenance, fetal growth, and maternal adaptation [5-7]. Among these hormones, human chorionic gonadotropin (hCG) plays a central role in early pregnancy by maintaining corpus luteum function and promoting progesterone production. Beyond its luteotropic effect, hCG regulates trophoblast differentiation, invasion, angiogenesis, and immune tolerance at the maternal–fetal interface [8-9]. Alterations in hCG production or bioactivity may therefore reflect abnormal placental development. Alpha-fetoprotein (AFP) is another key biochemical marker synthesized initially by the yolk sac and later predominantly by the fetal liver. AFP is transported into the maternal circulation via the placenta and fetal membranes, making maternal serum AFP (MS-AFP) a well-established second-trimester screening marker [10-11]. Traditionally, MS-AFP has been used for the detection of open neural tube defects and certain chromosomal abnormalities. However, accumulating evidence suggests that unexplained elevations in second-trimester MS-AFP may also reflect placental pathology, particularly disorders associated with abnormal placental adherence or vascular damage [12-14].
Pathological placentation is characterized by defective decasualization and abnormal trophoblast invasion. In placenta previa, implantation occurs in the lower uterine segment, increasing the risk of antepartum hemorrhage and adverse perinatal outcomes. More severe forms, grouped under the placenta accreta spectrum, involve abnormal adherence of chorionic villi to the myometrium due to partial or complete absence of the decidua basalis. These conditions are associated with massive obstetric hemorrhage, need for peripartum hysterectomy, and increased maternal mortality [1-3]. Despite advances in ultrasound and magnetic resonance imaging, a significant proportion of cases remain undiagnosed antenatally, underscoring the need for adjunctive biomarkers. Second-trimester biochemical markers offer a practical, widely available, and relatively inexpensive approach for risk stratification. During mid-gestation, placental mass, vascularity, and endocrine activity increase substantially. Deviations in maternal serum concentrations of placental-derived proteins during this period may therefore signal abnormal placental implantation or function. Elevated hCG levels in the second trimester have been associated with placental dysfunction, fetal aneuploidy, and adverse pregnancy outcomes, suggesting that altered trophoblastic activity persists beyond early gestation [8,9]. Similarly, increased MS-AFP levels may reflect enhanced leakage of fetal proteins into maternal circulation secondary to placental vascular disruption or abnormal placental adherence [12-14]. From a pathophysiological perspective, abnormal placentation shares features with invasive processes observed in malignancy, including dysregulated cell invasion, angiogenesis, and immune modulation. Both hCG and AFP are closely linked to trophoblast biology and placental integrity. HCG acts as an autocrine and paracrine regulator of trophoblast differentiation and syncytialization, while AFP levels may rise in response to increased placental permeability or tissue injury [9-14]. Thus, combined assessment of these markers may enhance the prediction of pathological placentation compared with imaging alone. The second trimester represents an optimal window for biochemical screening, as placental development has progressed sufficiently for abnormalities to manifest, while clinical complications may not yet be apparent. Identifying high-risk pregnancies during this stage allows referral to tertiary centers, multidisciplinary planning, and individualized timing and mode of delivery. Importantly, both hCG and AFP assays are already integrated into routine prenatal screening programs, facilitating potential translation into clinical practice without substantial additional cost or infrastructure [10,13]. In light of these considerations, evaluating second-trimester serum hCG and AFP as predictors of pathological placentation is of significant clinical relevance. Understanding the association between abnormal levels of these markers and placental disorders may improve antenatal detection, optimize maternal and fetal outcomes, and reduce the burden of obstetric morbidity. Therefore, this study aims to assess the relationship between second-trimester maternal serum hCG and AFP levels, measured between 24 and 30+6 weeks of gestation, and the risk of pathological placentation. The aim of this study was to assess the association between 2nd trimester level of serum hCG and serum AFP measured between 24-30+6 weeks of gestation in predicting pathological placentation.
Pateints and Methods
This Prospective Case-Control study was conducted in Tikrit City from the period: 1st of December 2023 to 1st of September 2024.
Inclusion Criteria
The study included 90 pregnant women with singleton gestations who delivered at Tikrit Teaching Hospital and had complete documentation of second-trimester serum screening results. Eligible participants were those who underwent maternal serum biochemical screening between the 24 and 30+6 weeks of gestation. The study cohort was divided into the following groups:
Women with Pathological Presentation (n:60)
Control Group
Women with normal placentation (n:30). Controls were matched with cases based on gestational age at delivery, parity, and history of previous cesarean sections.
Exclusion Criteria
Multiple gestations (e.g., twins, triplets)
Women with incomplete or missing serum screening data
Presence of congenital anomalies or chromosomal abnormalities in the fetus
Women with other significant comorbidities that could independently affect pregnancy outcomes (e.g., pre-eclampsia, chronic hypertension, diabetes mellitus)
Cases where gestational age could not be accurately determined due to lack of reliable last menstrual period data or ultrasound confirmation
History of maternal conditions that affect placental function unrelated to pathological placentation, such as thrombophilias or autoimmune disorders (systemic lupus erythematous)
Administrative Approvals were Granted from the Following
Scientific council of obstetrics and gynecology / iraqi board for medical specialization
Authorization for the study was granted by the Director of the Tikrit Health Directorate
All participants were interviewed using a questionnaire developed by the investigator to collect relevant data
Data collection
Data were retrieved from the comprehensive computerized perinatal database of the obstetric division and the ambulatory database. Each patient was assigned a unique identification number to ensure accurate cross-tabulation of data. Collected data included maternal demographics (e.g., age), obstetric parameters (e.g., parity), risk factors for pathological placentation (e.g., history of previous uterine surgical procedures), maternal and neonatal outcomes (e.g., hysterectomy, NICU admission), and values of second-trimester biochemical markers (AFP and hCG).