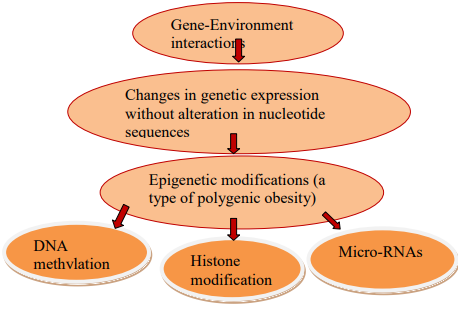
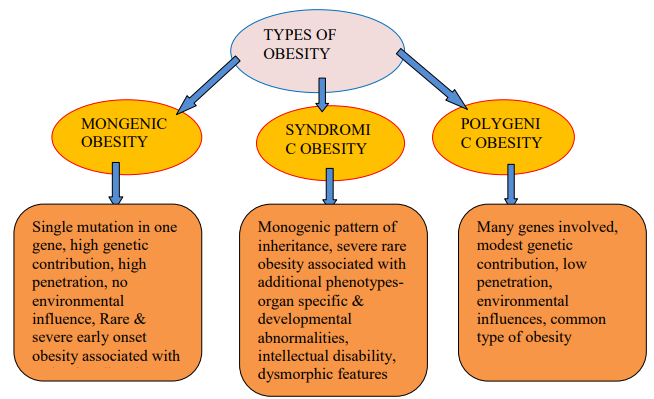
Lee, Y.C. et al. “Using Machine Learning to Predict Obesity Based on Genome-Wide and Epigenome-Wide Gene–Gene and Gene–Diet Interactions.” Frontiers in Genetics, vol. 12, 2022, p. 783845.
Loos, R.J. and G.S. Yeo. “The Genetics of Obesity: From Discovery to Biology.” Nature Reviews Genetics, vol. 23, no. 2, 2022, pp. 120–133.
Atoum, M.F. “Association of Leptin Receptor Q223R Gene Polymorphism and Breast Cancer Patients: A Case Control Study.” Asian Pacific Journal of Cancer Prevention: APJCP, vol. 23, no. 1, 2022, p. 177.
Marotta, V. et al. “Exposure to Bisphenol A Increases Malignancy Risk of Thyroid Nodules in Overweight/Obese Patients.” Environmental Pollution, vol. 316, 2023, p. 120478.
Tanofsky-Kraff, M. et al. “The FTO Gene rs9939609 Obesity-Risk Allele and Loss of Control over Eating.” The American Journal of Clinical Nutrition, vol. 90, no. 6, 2009, pp. 1483–1488.
Dashti, H.S. et al. “Interaction of Obesity Polygenic Score with Lifestyle Risk Factors in an Electronic Health Record Biobank.” BMC Medicine, vol. 20, 2022, pp. 1–12.
Zou, X. et al. “Deciphering the Irregular Risk of Stroke Increased by Obesity Classes: A Stratified Mendelian Randomization Study.” Frontiers in Endocrinology, vol. 12, 2021, p. 750999.
Wang, Y. et al. “MC4R Gene Polymorphisms Interact with the Urbanized Living Environment on Obesity: Results from the Yi Migrant Study.” Frontiers in Genetics, vol. 13, 2022, p. 849138.
Wang, J. et al. “Genetic Predisposition of Both Waist Circumference and Hip Circumference Increased the Risk of Venous Thromboembolism.” Thrombosis and Haemostasis, vol. 123, no. 3, 2023, pp. 347–361.
Sabir, J.S. et al. “The Genetic Association Study of TP53 Polymorphisms in Saudi Obese Patints.” Saudi Journal of Biological Sciences, vol. 26, no. 7, 2019, pp. 1338–1343.
Thaker, V.V. “Genetic and Epigenetic Causes of Obesity.” Adolescent Medicine: State of the Art Reviews, vol. 28, no. 2, 2017, p. 379.
Ling, C. and T. Rönn. “Epigenetics in Human Obesity and Type 2 Diabetes.” Cell Metabolism, vol. 29, no. 5, 2019, pp. 1028–1044.
Ansari, Y. et al. “Association Study of Melanocortin-4 Receptor (rs17782313) and PKHD1 (rs2784243) Variations and Early Incidence of Obesity at the Age of Maturity.” Advances in Human Biology, vol. 13, no. 1, 2023, pp. 144–150.
Aboonq, M.S. et al. “Methylenetetrahydrofolate Reductase and Peroxisome Proliferator-Activated Receptor-γ Gene Polymorphisms and Risk of Obesity in Females of Madina, Saudi Arabia.” Bulletin of Egyptian Society for Physiological Sciences, vol. 43, no. 1, 2023, pp. 64–74.
Chauhan, G. et al. “Common Variants of FTO and the Risk of Obesity and Type 2 Diabetes in Indians.” Journal of Human Genetics, vol. 56, no. 10, 2011, pp. 720–726.
Prakash, J. et al. “Association of FTO rs9939609 SNP with Obesity and Obesity-Associated Phenotypes in a North Indian Population.” Oman Medical Journal, vol. 31, no. 2, 2016, p. 99.
Younes, S. et al. “Genetic Polymorphisms Associated with Obesity in the Arab World: A Systematic Review.” International Journal of Obesity, vol. 45, no. 9, 2021, pp. 1899–1913.
Khatami, M. et al. “Relationship of the MTHFD1 (rs2236225), eNOS (rs1799983), CBS (rs2850144) and ACE (rs4343) Gene Polymorphisms in a Population of Iranian Pediatric Patients with Congenital Heart Defects.” The Kaohsiung Journal of Medical Sciences, vol. 33, no. 9, 2017, pp. 442–448.
Ren, Q. et al. “Impact of Variants of the EXT2 Gene on Type 2 Diabetes and Its Related Traits in the Chinese Han Population.” Endocrine Research, vol. 40, no. 2, 2015, pp. 79–82.
Liu, G.C. et al. “Interaction between TP53 and XRCC1 Increases Susceptibility to Cervical Cancer Development: A Case Control Study.” BMC Cancer, vol. 19, 2019, pp. 1–9.
Li, L. et al. “Exercise Retards Ongoing Adipose Tissue Fibrosis in Diet-Induced Obese Mice.” Endocrine Connections, vol. 10, no. 3, 2021, pp. 325–335.
Fu, X. et al. “Association between PRDM16, MEF2D, TRPM8, LRP1 Gene Polymorphisms and Migraine Susceptibility in the She Ethnic Population in China.” Clinical and Investigative Medicine, vol. 42, no. 1, 2019, pp. E21–E30.
Srivastav, A. et al. “Genetics of Obesity.” Indian Journal of Clinical Biochemistry, vol. 31, no. 4, 2016, pp. 361–371.
Kasinska, M.A. et al. “Epigenetic Modifications in Adipose Tissue–Relation to Obesity and Diabetes.” Archives of Medical Science, vol. 12, no. 6, 2016, pp. 1293–1301.
Ali, Mohamed M. et al. “DNA Methylation Profile of Genes Involved in Inflammation and Autoimmunity Correlates with Vascular Function in Morbidly Obese Adults.” Epigenetics, 2021. https://doi.org/10.1080/15592294.2021.1876285.
Leija-Martínez, J.J. et al. “Promoter Methylation Status of RORC, IL17A, and TNFA in Peripheral Blood Leukocytes in Adolescents with Obesity-Related Asthma.” Heliyon, vol. 8, no. 12, 2022.
Lima, R.P.A. et al. “Effect of a Diet Containing Folate and Hazelnut Oil Capsule on the Methylation Level of the ADRB3 Gene, Lipid Profile and Oxidative Stress in Overweight or Obese Women.” Clinical Epigenetics, vol. 9, 2017, pp. 1–9.
Hammond, C.M. et al. “Histone Chaperone Networks Shaping Chromatin Function.” Nature Reviews Molecular Cell Biology, vol. 18, no. 3, 2017, pp. 141–158.
Bricambert, J. et al. “Impaired Histone Deacetylases 5 and 6 Expression Mimics the Effects of Obesity and Hypoxia on Adipocyte Function.” Molecular Metabolism, vol. 5, no. 12, 2016, pp. 1200–1207.
Kang, H.S. et al. “IDH1-Dependent α-KG Regulates Brown Fat Differentiation and Function by Modulating Histone Methylation.” Metabolism, vol. 105, 2020, p. 154173.
Chen, L. et al. “MiR-146b Is a Regulator of Human Visceral Preadipocyte Proliferation and Differentiation and Its Expression Is Altered in Human Obesity.” Molecular and Cellular Endocrinology, vol. 393, nos. 1–2, 2014, pp. 65–74.
Ayeldeen, G. et al. “Signature of Micro RNA 146a/215 and IL-6/TGF-β Levels in a Cross-Link Axis between Obesity and Colorectal Cancer.” Non-Coding RNA Research, vol. 8, no. 2, 2023, pp. 187–191.
Bastos, R.V. et al. “Acute Green Tea Intake Attenuates Circulating MicroRNA Expression Induced by a High-Fat, High-Saturated Meal in Obese Women: A Randomized Crossover Study.” The Journal of Nutritional Biochemistry, vol. 112, 2023, p. 109203.
Lacedonia, D. et al. “Different Expression of Micro-RNA in the Subcutaneous and Visceral Adipose Tissue of Obese Subjects.” Rejuvenation Research, vol. 25, no. 2, 2022, pp. 89–94.
Man, S. et al. “Association between Metabolically Healthy Obesity and Non-Alcoholic Fatty Liver Disease.” Hepatology International, vol. 16, no. 6, 2022, pp. 1412–1423.
Pasqual, E. et al. “Obesity, Obesity-Related Metabolic Conditions, and Risk of Thyroid Cancer in Women: Results from a Prospective Cohort Study (Sister Study).” The Lancet Regional Health–Americas, vol. 23, 2023.
Jeanes, Y.M. and S. Reeves. “Metabolic Consequences of Obesity and Insulin Resistance in Polycystic Ovary Syndrome: Diagnostic and Methodological Challenges.” Nutrition Research Reviews, vol. 30, no. 1, 2017, pp. 97–105.
Chen, L. et al. “Association of Different Obesity Patterns with Hypertension in US Male Adults: A Cross-Sectional Study.” Scientific Reports, vol. 13, no. 1, 2023, p. 10551.
Kalaydzhiev, P. et al. “Sleep Apnea in Patients with Exacerbated Heart Failure and Overweight.” Sleep Medicine: X, vol. 5, 2023, p. 100065.
Glasstetter, L.M., et al. “Obesity and Dyslipidemia Are Associated with Partially Reversible Modifications to DNA Hydroxymethylation of Apoptosis-and Senescence-Related Genes in Swine Adipose-Derived Mesenchymal Stem/Stromal Cells.” Stem Cell Research & Therapy, vol. 14, no. 1, 2023, pp. 1–19.
Grotle, M. et al. “Obesity and Osteoarthritis in Knee, Hip and/or Hand: An Epidemiological Study in the General Population with 10 Years Follow-Up.” BMC Musculoskeletal Disorders, vol. 9, 2008, pp. 1–5.
Marrodan, M. et al. “Obesity and the Risk of Multiple Sclerosis. The Role of Leptin.” Annals of Clinical and Translational Neurology, vol. 8, no. 2, 2021, pp. 406–424.
Duan, D.M. et al.“Modification Effect of Sex and Obesity on the Correlation of LEP Polymorphisms with Leptin Levels in Taiwanese Obese Women.” Molecular Genetics & Genomic Medicine, vol. 8, no. 3, 2020, p. e1113.
Bouafi, H. et al. “Genetic Association of LEP Gene Polymorphisms with Obesity in Moroccan Individuals: Case–Control Study and Updated Meta-Analysis.” Biochemical Genetics, 2023, pp. 1–17.
Darwish, N.M. et al. “PPARG Expression Patterns and Correlations in Obesity.” Journal of King Saud University-Science, vol. 34, no. 6, 2022, p. 102116.
Milagro, F.I. et al. “CLOCK, PER2 and BMAL1 DNA Methylation: Association with Obesity and Metabolic Syndrome Characteristics and Monounsaturated Fat Intake.” Chronobiology International, vol. 29, no. 9, 2012, pp. 1180–1194.
Shen, J. and B. Zhu. “Integrated Analysis of the Gene Expression Profile and DNA Methylation Profile of Obese Patients with Type 2 Diabetes.” Molecular Medicine Reports, vol. 17, no. 6, 2018, pp. 7636–7644.
Longo, M. et al. “Epigenetic Modifications of the Zfp/ZNF423 Gene Control Murine Adipogenic Commitment and Are Dysregulated in Human Hypertrophic Obesity.” Diabetologia, vol. 61, 2018, pp. 369–380.
Houde, A.A. et al.“Leptin and Adiponectin DNA Methylation Levels in Adipose Tissues and Blood Cells Are Associated with BMI, Waist Girth and LDL-Cholesterol Levels in Severely Obese Men and Women.” BMC Medical Genetics, vol. 16, 2015, pp. 1–10.
Castellano-Castillo, D. et al. “Genome Profiling of H3k4me3 Histone Modification in Human Adipose Tissue during Obesity and Insulin Resistance.” Biomedicines, vol. 9, no. 10, 2021, p. 1363.
Tanofsky-Kraff, M. et al. “The FTO Gene rs9939609 Obesity-Risk Allele and Loss of Control over Eating.” The American Journal of Clinical Nutrition, vol. 90, no. 6, 2009, pp. 1483–1488.
Dashti, H.S. et al. “Interaction of Obesity Polygenic Score with Lifestyle Risk Factors in an Electronic Health Record Biobank.” BMC Medicine, vol. 20, 2022, pp. 1–12.
Pillon, N.J. et al. “Metabolic Consequences of Obesity and Type 2 Diabetes: Balancing Genes and Environment for Personalized Care.” Cell, vol. 184, no. 6, 2021, pp. 1530–1544.
Lazarus, E. and H.E. Bays. “Cancer and Obesity: An Obesity Medicine Association (OMA) Clinical Practice Statement (CPS) 2022.” Obesity Pillars, vol. 3, 2022, p. 100026.
Zou, X. et al. “Deciphering the Irregular Risk of Stroke Increased by Obesity Classes: A Stratified Mendelian Randomization Study.” Frontiers in Endocrinology, vol. 12, 2021, p. 750999.
Lundgren, S. et al. “BMI Is Positively Associated with Accelerated Epigenetic Aging in Twin Pairs Discordant for BMI.” MedRxiv, 2021, p. 2021-03.
Ayeldeen, G. et al. “Signature of Micro RNA 146a/215 and IL-6/TGF-β Levels in a Cross-Link Axis between Obesity and Colorectal Cancer.” Non-Coding RNA Research, vol. 8, no. 2, 2023, pp. 187–191.
Wang, Y. et al. “MC4R Gene Polymorphisms Interact with the Urbanized Living Environment on Obesity: Results from the Yi Migrant Study.” Frontiers in Genetics, vol. 13, 2022, p. 849138.
Willmer, T. et al. “DNA Methylation of FKBP5 in South African Women: Associations with Obesity and Insulin Resistance.” Clinical Epigenetics, vol. 12, no. 1, 2020, pp. 1–15.