The following results were obtained from the physic-chemical parameters monitored during the study period:
pH in surface water: The results of the analysis of pH in surface water is presented in appendix 1 and represented in fig 1. It ranged from 6.2 to 7.4 at the 5 stations sampled during the sampling period. The mean values recorded during the period is station 1 (6.8), station 2 (7.1), station 3 (6.8), station 4(6.9) and station 5(6.85).
Seasonally, dry season (6.91) was slightly higher than the rainy season (6.90).
The analysis of variance (ANOVA) results presented in appendix 5 shows that there is significant difference (p<0.05) between location and time for pH values obtained during the monitoring period (P = 0.009).
ORP in surface water: The results of analysis obtained for ORP is presented in fig 2 and range from 97.2 to 134.8 with the highest ORP in the month of September (208.3).
Seasonally, dry season (119) is higher than rainy season (112).
The analysis of variance (ANOVA) results show that there is significant difference at P< 0.05 between record obtained in the rest of the months and stations sampled during the period. P=0.002.
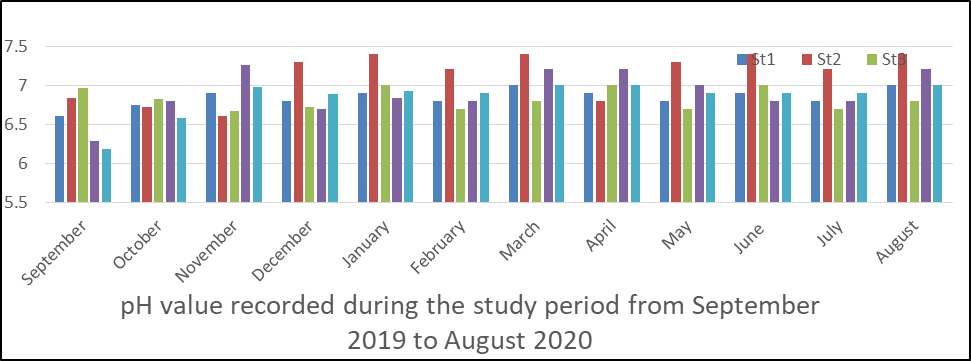
Fig 1 pH in water sample
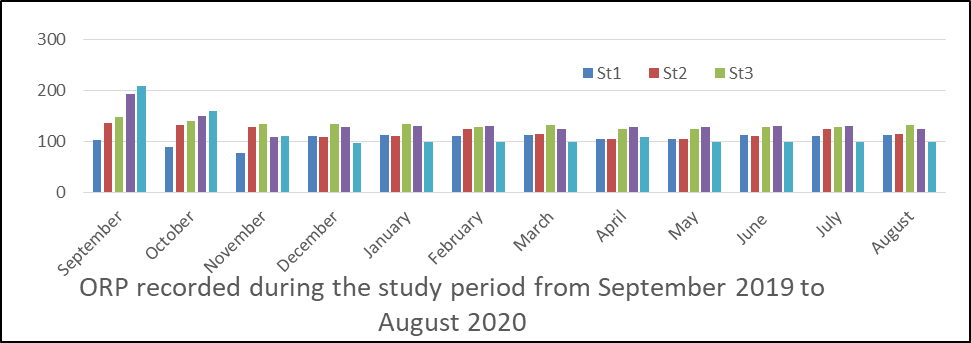
Fig 2 ORP in surface water
DO in surface water
Fig 3 shows the range for dissolved oxygen (DO) to be 3.28 to 5.69. The mean values were station 1(4.28), station 2(4.53), station 3(5.06), station 4(4.83) and station 5(4.88). The highest levels occur in September (5.69) in station 2 followed by station 3 (5.67).
The seasonal and spatial variations in DO as shown in fig 3 revealed that dry season (5.79) is higher than rainy season (4.75).
The analysis of variance result shows that there is no significant difference p< 0.05 between the record obtained in the month and station during the period p=0.057
BOD in surface water
The result obtained for BOD is presented in fig 4 and the value ranged from 1.13 to 4.14. The mean level obtained were station 1(2.74), station 2(2.80), station 3(3.13), station 4(2.88) and station 5(2.83).
Seasonally, the mean value for rainy season (3.17) is higher than that obtained for dry season (2.65).
In the analysis of variance (ANOVA) there is no significant difference (p< 0.05) for BOD in the water between time and location during the study period (P=0. 337).
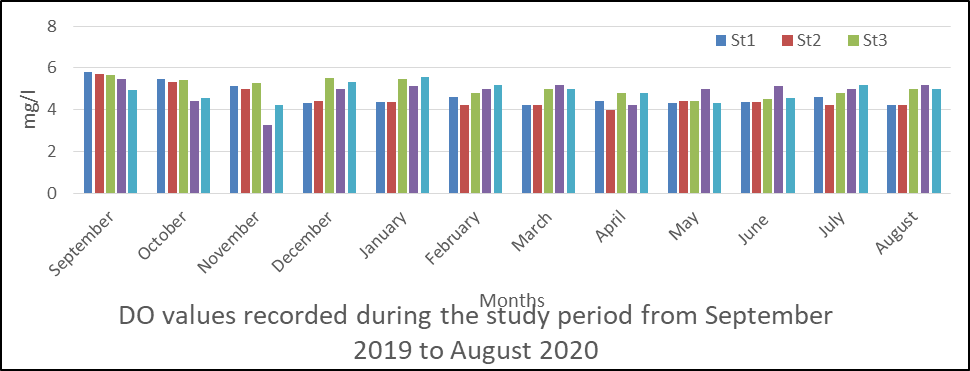
Figs 3 DO in surface water
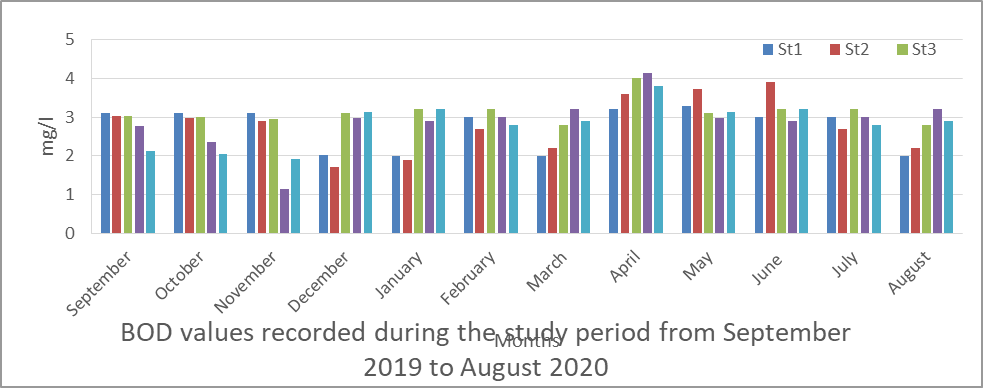
Fig 4 BOD in surface water
Conductivity in surface water
The result of analysis obtained for conductivity is presented in fig 5 and the value ranged from 11to122. The mean values for conductivity measurements were station 1(81), station 2(59.3), station 3(41.9), station 4(45.4) and station 5(56.9).
The seasonal and spatial variation shows a higher value in the rainy season (59.8) than the dry season (55.7).
The analysis of variance (ANOVA), indicated that there was significant difference between time (month) at p<0. 05 for conductivity value obtained during the monitoring period. P=0.042.
Salinity in surface water
The result of analysis obtained for salinity is presented in fig 6 and ranged from 0.0 6 to 0.09mg/l. The mean values for salinity measurements were station 1(0.025), station 2(0.0125), station 3(0.0125), station 4(0.03) and station 5(0.02mg/l). The measurement according to months is also shown in fig 6 and recorded little variations with the highest values in January, February, in the five stations sampled.
The analysis of variance (ANOVA) indicated that there was no significant difference at p <0.05 for salinity values obtained during the monitoring period P=0.887.
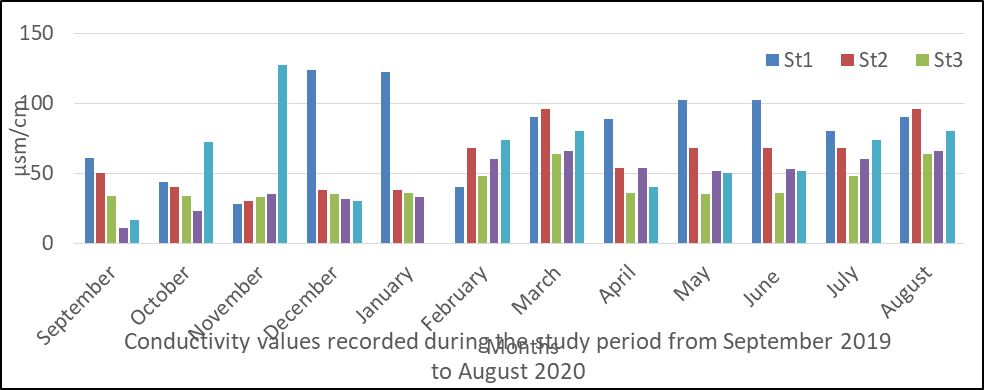
Fig 5: Conductivity in surface water
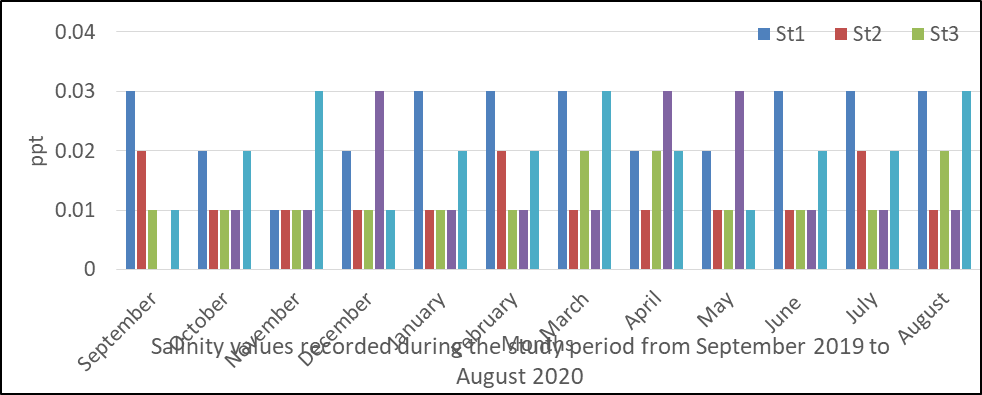
Fig 6: Salinity in surface water
TDS in surface water
The result of analysis obtained for TDS is presented in fig 7 and ranged from 6 to 64mg/l.
The mean values for TDS measurements were station 1(28.57), station 2(24.03), station 3(17.55), station 4(4.99) and station 5(22.35mg/l).
The measurement according to months is also shown in fig 7 and recorded the highest values in December and February in the five stations sampled.
The analysis of variance (ANOVA) indicated that there was no significant difference between time (month) at p <0.05 for TDS values obtained during the monitoring period P=0.825.
Turbidity in surface water
The result of analysis obtained for turbidity is presented in fig 8 and ranged from 13.5 to 61.6mg/l during the monitoring period.
The mean values are station 1(24.89), station 2(27.38), station 3(27.32), station 4(22.94) and station 5(23.34mg/l).
The result of the variations shows that rainy season had higher mean value than dry season. The highest turbidity value recorded during the period occurred in September.
The analysis of variance (ANOVA) results shows that there is no significant difference (P <0.05) between the values recorded for each water sample over time during the monitoring period (P=0.056).
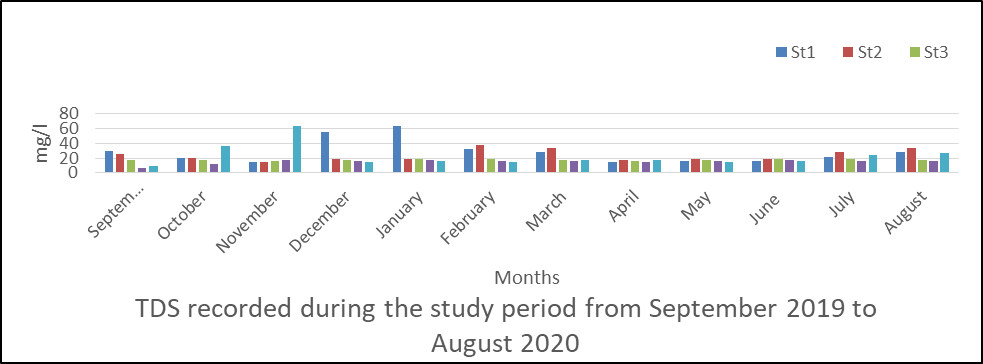
Fig 7: TDS in surface water
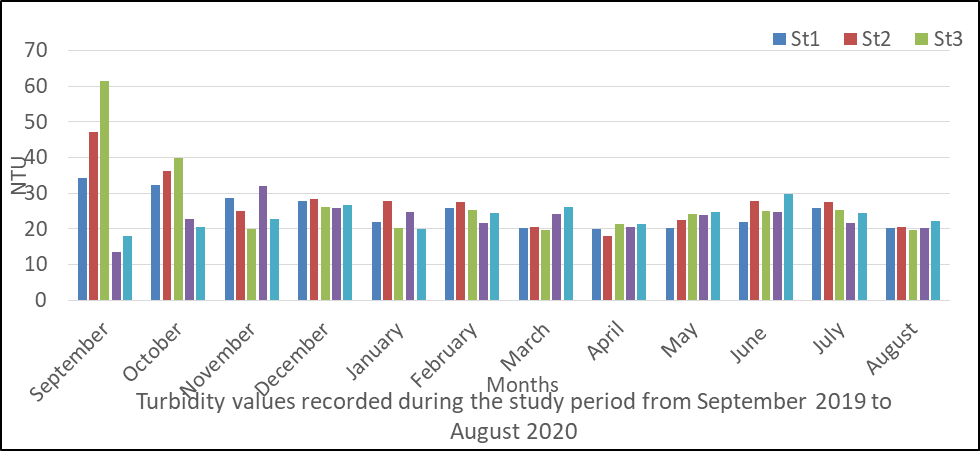
Fig 8: Turbidity in surface water
Temperature in Surface Water
The spatial changes in temperature are presented in fig 9. Generally there was fluctuation in temperature values of the river water during the monitoring period and ranged from 28.89 to 32.4. The mean values are station 1(29.78), station 2(30.87), station 3(28.97), station 4(30.29) and station 5(29.98oC).
The result of the variations shows that dry season (32.4) had higher mean value than rainy season (27.7).
The analysis of variance (ANOVA) results shows that there was significant difference (P <0.05) in the temperature values obtained during the monitoring period (P=0.056).
Phosphate in Surface Water
Fig 10 shows the result of phosphate levels in water which ranged from 0.64 to 2.4. The specific mean values were station 1(0.815), station 2(1.03), station 3(0.785), station 4(0.177) and station 5(1.27).
Seasonally, phosphate was observed throughout the monitoring period but the rainy season had higher mean value compared to the dry season.
The analysis of variance (ANOVA) results shows that there was significant difference (P <0.05) in phosphate values obtained during the monitoring period (P=0.001).
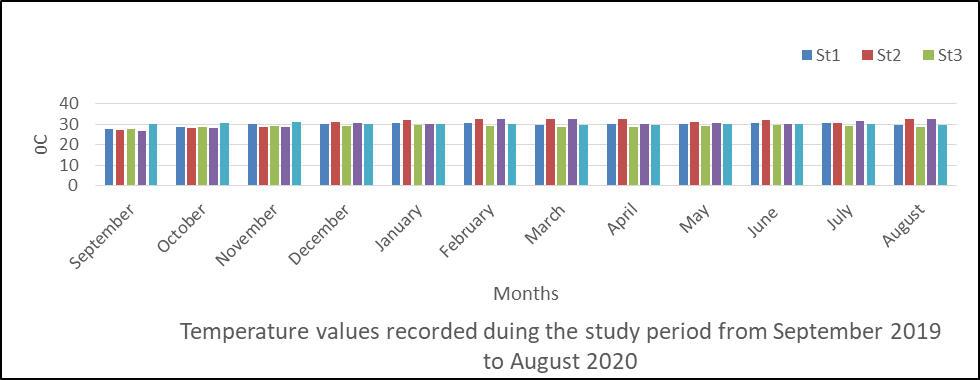
Fig 9: Temperature in surface water
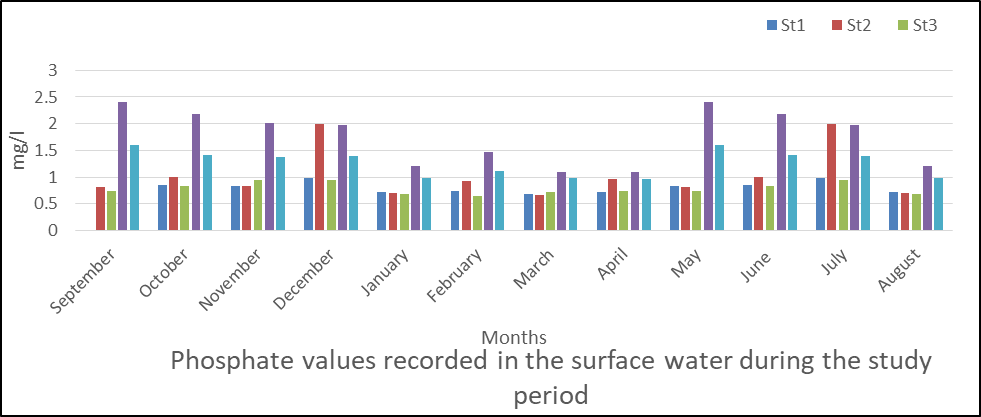
Fig 10: Phosphate in surface water
Sulphate in Surface Water
The results obtained in sulphate are presented in fig 11 and sulphate concentration ranged from 1 to 2.52mg/l. The mean values are station 1(1.83), station 2(1.95), station 3(1.29), station 4(1.88) and station 5(1.04).
For seasonal variation, rainy season tends to be higher than dry season in the five stations sampled. Fig 11 also shows the concentration in months with highest values in September and May followed by November.
The analysis of variance (ANOVA) shows significant difference (p < 0.05) in sulphate values of the water samples during the sampling period (P=0,000).
Nitrate in Surface Water
The results of the analysis obtained for nitrate in water is presented in fig 12 with the nitrate values ranged from 0.61 to 1.32mg/l. The mean values are station 1(0.73), station 2(0.78), station 3(0.88), station 4(0.96) and station 5(1.04mg/l).
Seasonally, the rainy season recorded a higher mean value than the dry season.
The analysis of variance shows that there was significant difference (P< 0.05) in nitrate values of water samples during the sampling period. (P=0.000).
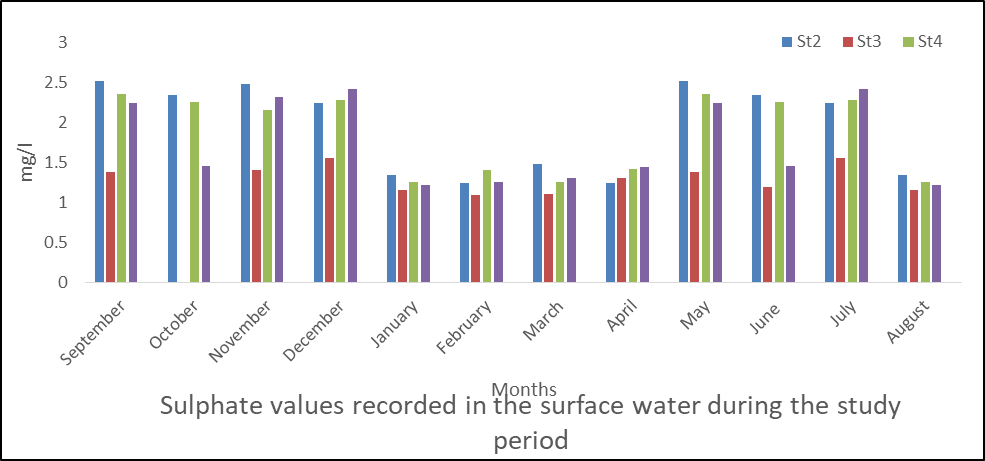
Fig 11: Sulphate in surface water
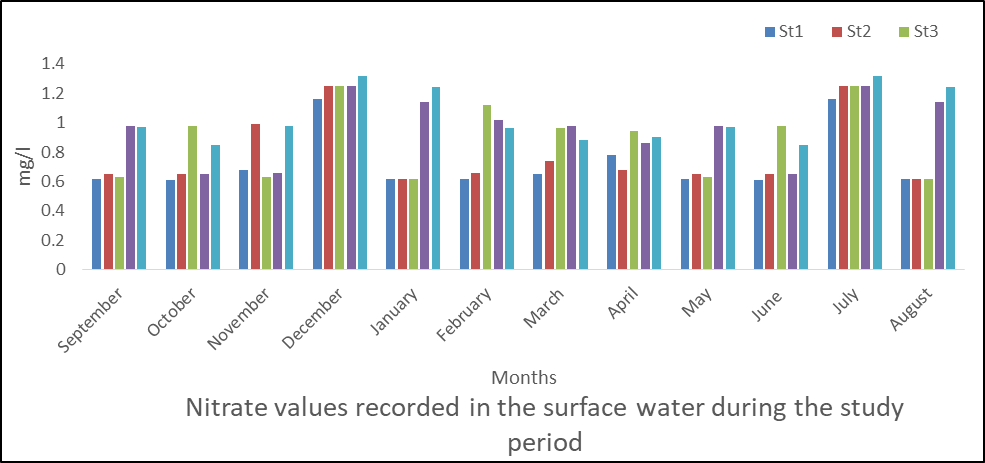
Fig 12: Nitrate in surface water
Available Phosphorus (AVP) in Sediment
Fig 13 shows the result of AVP in sediment which ranges from 0.35 to 3.98mg/kg. The specific mean values were station1 (2.25), station 2(3.08), station 3(3.12), station 4(3.56) and station 5(2.50). These values could be considered moderate and slightly below the permissible limit of 5mg/kg (FMENV, 2001).
Seasonally, the rainy season recorded a higher phosphate level than the dry season
The analysis of variance (ANOVA) shows that there is significant difference (P< 0.05) both in location and time (P=0.000).
Nitrate in Sediments
The result of nitrate levels in sediment is presented in Fig 14 with the values ranging from 0.825 to 3.64mg/kg. The mean values are station 1(1.65), station 2(1.80), station 3(2.90), station 4(1.92) and station 5(2.12). These values are within the permissible limit of 5mg/kg (FMEnv, 2001) [1].
Seasonally, rainy season recorded the highest value as compared to values recorded for dry season.
The result of analysis of variance (ANOVA) showed that there is significant difference (p< 0.05) between locations and time (p= 0.000).
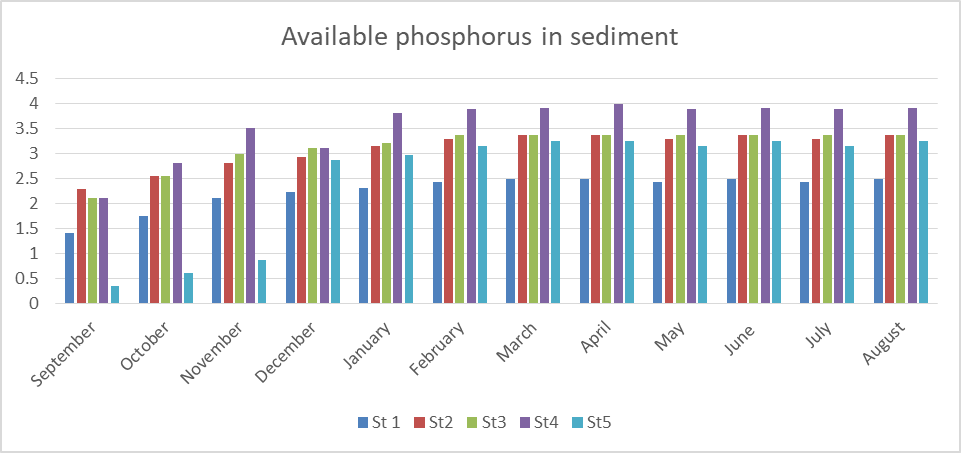
Fig 13: Phosphate in sediment
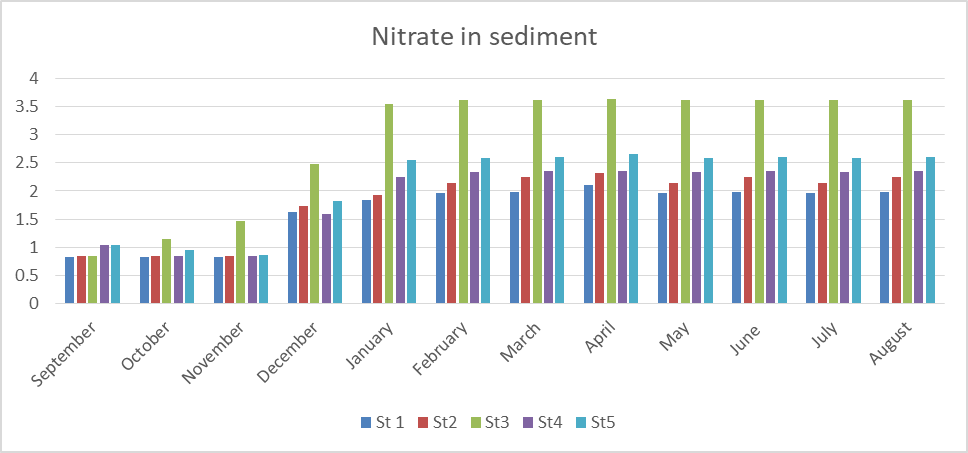
Fig 14: Nitrate in Sediment
Sulphate in Sediment
Fig 15 shows the result of sulphate in sediment which ranges from 0.7- 6.12mg/kg. The specific mean values were station 1(3.42), station 2(3.05), station 3(2.34), station 4(5.51) and station 5(2.05). These results indicate moderate level of sulphate in accordance with the FMENV (1999) permissible level in sediment of < 5mg/kg.
Seasonally, the rainy season recorded a higher sulphate level than the dry season in the 5 stations sampled. The highest value was observed in April in all the stations.
The analysis of variance (ANOVA) shows that there is significant difference (P< 0.05) both in location and time (P=0.002).
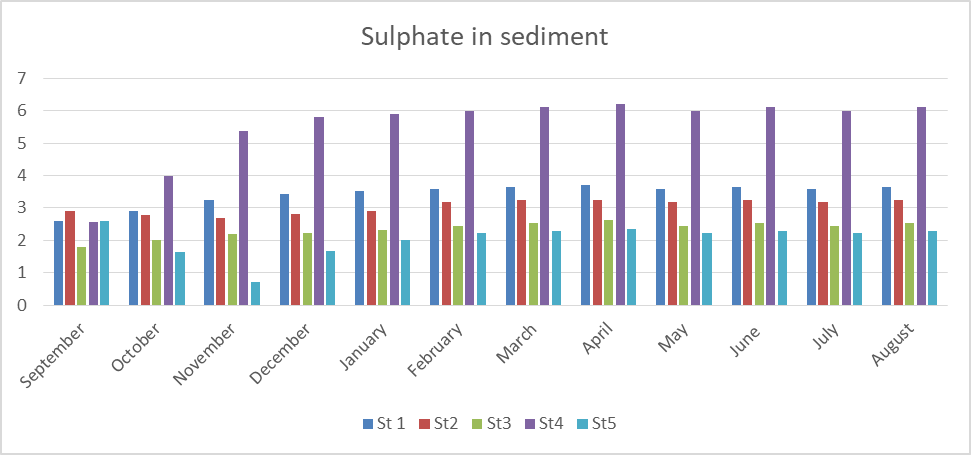
Fig15: Sulphate in Sediment
















 conc.
conc.
